Online Database of Chemicals from Around the World
| Jinxiang Chemical (Danyang) Products Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinchemical.com | |||
 | +86 (25) 8681-9862 +86 13913839793 | |||
 | +86 (25) 8681-9859 | |||
 | sales@jinchemical.com nancy-zong@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Junkai (Tianjin) Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.junkaichem.com | |||
 | +86 (22) 8568-9515 ex 8818 +86 13920864700 | |||
 | +86 (22) 2378-7875 | |||
 | sales@junkaichem.com jane-jkchem@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Synasia Inc | USA | |||
|---|---|---|---|---|
 | www.synasia.com | |||
 | +1 (732) 205-9880 | |||
 | +1 (732) 205-1788 | |||
 | info@synasia.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Valiant Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.valiant-cn.com | |||
 | +86 (535) 610-1878 | |||
 | +86 (535) 638-6989 | |||
 | jiangyanan@valiant-cn.com chenxiaowei@valiant-cn.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chengdu Yuanda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ydachem.com | |||
 | +86 (28) 8535-6300 | |||
 | +86 (28) 8535-6300 | |||
 | sales@ydachem.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Guangzhou Taiji New Materials Co., Ltd.. | China | |||
|---|---|---|---|---|
 | www.taijichem.com | |||
 | +86 18620003222 | |||
 | +86 (20) 8755-3522 | |||
 | tailuck@qq.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2019 | ||||
| CHINATECH (TIANJIN) CHEMICAL CO., LTD. | China | |||
|---|---|---|---|---|
 | www.chinatechem.com | |||
 | +86 (22) 8372-6202 EXT8819 | |||
 | sales4@chinatechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Wuhan Comings Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | comings.en.made-in-china.com | |||
 | +86 13659820406 | |||
 | info@comingschemi.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Wuxi Cidic Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cidic.com.cn | |||
 | +86 (510) 8230-3030 | |||
 | +86 (510) 8230-3031 | |||
 | baoming@cidic.com.cn | |||
| Chemical distributor | ||||
| Classification | Organic raw materials >> Aryl compounds >> Anilines |
|---|---|
| Name | 4,4'-(4,4'-Isopropylidenediphenyl-1,1'-diyldioxy)dianiline |
| Synonyms | 2,2-Bis[4-(4-aminophenoxy)phenyl]propane |
| Molecular Structure | 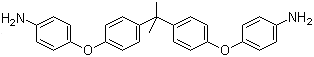 |
| Molecular Formula | C27H26N2O2 |
| Molecular Weight | 410.51 |
| CAS Registry Number | 13080-86-9 |
| EC Number | 235-985-8 |
| SMILES | CC(C)(C1=CC=C(C=C1)OC2=CC=C(C=C2)N)C3=CC=C(C=C3)OC4=CC=C(C=C4)N |
| Density | 1.2$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 127 - 130 $degree$C (Expl.) |
| Boiling point | 587.1$+/-$50.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 322.8$+/-$23.8 $degree$C (Calc.)* |
| Index of refraction | 1.641 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H319-H335-H413 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P272-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
4,4'-(4,4'-Isopropylidenediphenyl-1,1'-diyldioxy)dianiline is an aromatic diamine compound in which two para-aminophenyl groups are connected through ether linkages to a central 4,4'-isopropylidenediphenyl unit. This compound is a multifunctional monomer widely used in the synthesis of high-performance polymers, particularly polyimides and epoxy resins. Its symmetrical structure, rigid aromatic core, and reactive amino groups make it a valuable building block for materials requiring thermal stability, mechanical strength, and chemical resistance. The molecular structure features a central bisphenol-like moiety, where the two phenyl rings are connected via an isopropylidene bridge, forming a 4,4'-isopropylidenediphenyl unit. This core is linked to two para-aminophenoxy groups, providing a total of two primary amine functionalities. The combination of ether linkages and the bulky isopropylidene unit introduces flexibility in processing while maintaining rigidity in the polymer backbone. The amino groups are highly reactive toward electrophiles, enabling polymerization and crosslinking reactions. Synthesis of this compound typically involves the nucleophilic substitution of bisphenol A derivatives with protected aminophenol units, followed by deprotection to yield the free amino groups. Such strategies ensure the proper placement of functional groups and preservation of the symmetrical structure, producing a high-purity monomer suitable for polymer applications. In polymer chemistry, 4,4'-(4,4'-isopropylidenediphenyl-1,1'-diyldioxy)dianiline serves as a diamine monomer for polyimide production. Reaction with aromatic dianhydrides produces polyimides with excellent thermal stability, mechanical properties, and chemical resistance. The isopropylidene group contributes to reduced chain packing and increased free volume, improving solubility and processability compared to fully aromatic diamines. These characteristics are important in electronic, aerospace, and coating applications. The compound’s amino groups also allow its use in epoxy resins, where they react with epoxide groups to form highly crosslinked thermosetting networks. The combination of rigid aromatic segments and reactive sites produces resins with high glass transition temperatures, low moisture absorption, and strong adhesion properties. Such materials are widely applied in adhesives, coatings, and advanced composites. Beyond polymer synthesis, this diamine serves as an intermediate in organic synthesis for functional derivatives. Its reactive amino groups can undergo acylation, alkylation, or condensation reactions, enabling the preparation of specialized molecules and functional materials. The bulky isopropylidene core influences steric properties and reaction selectivity, which is advantageous in designing complex molecules. Overall, 4,4'-(4,4'-isopropylidenediphenyl-1,1'-diyldioxy)dianiline is a multifunctional aromatic diamine that combines reactive amino groups, a rigid central core, and ether linkages. These features make it an essential monomer for high-performance polymers and functional materials requiring thermal stability, mechanical strength, and controlled physical properties. References 2025. A review of recent development in preparation and modification of waterborne polyurethanes and their application in waterborne inks for plastic packaging printing. Journal of Coatings Technology and Research. DOI: 10.1007/s11998-024-01072-6 2025. Exploring the applicative potential of polyimides containing DOPO-functionalized triazine through straightforward blending technique. Journal of Materials Science. DOI: 10.1007/s10853-025-10751-7 2024. Preparation of Super Slippery Surface of Polyimide Composite Microspheres. Polymer Science, Series B. DOI: 10.1134/s1560090424601146 |
| Market Analysis Reports |