Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Biochemical >> Inhibitor >> Epigenetics >> HDAC inhibitor |
|---|---|
| Name | Tucidinostat |
| Synonyms | N-(2-amino-4-fluorophenyl)-4-[[[(E)-3-pyridin-3-ylprop-2-enoyl]amino]methyl]benzamide |
| Molecular Structure | 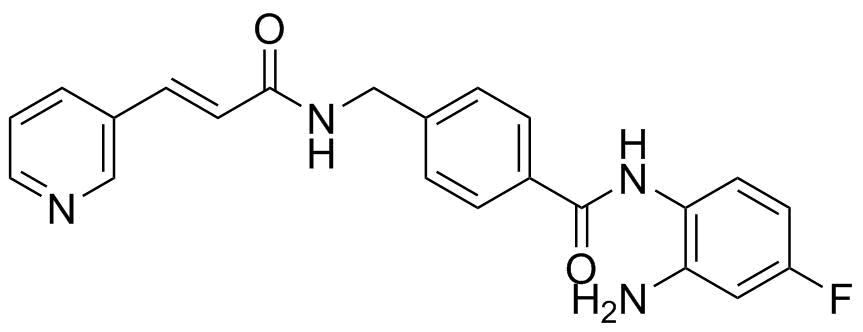 |
| Molecular Formula | C22H19FN4O2 |
| Molecular Weight | 390.41 |
| CAS Registry Number | 1616493-44-7 |
| SMILES | C1=CC(=CN=C1)/C=C/C(=O)NCC2=CC=C(C=C2)C(=O)NC3=C(C=C(C=C3)F)N |
| Density | 1.3$+/-$0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.691, Calc.* |
| Boiling Point | 602.1$+/-$55.0 $degree$C (760 mmHg), Calc.* |
| Flash Point | 317.9$+/-$31.5 $degree$C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Tucidinostat is an orally administered anticancer drug that belongs to the class of histone deacetylase (HDAC) inhibitors. It is used primarily in the treatment of certain hematological malignancies and works by modulating epigenetic regulation of gene expression rather than directly targeting DNA mutations. By inhibiting HDAC enzymes, tucidinostat alters chromatin structure and influences transcriptional activity, leading to growth arrest, differentiation, and apoptosis of cancer cells. The development of tucidinostat arose from research into epigenetic mechanisms involved in cancer progression. Histone deacetylases are enzymes that remove acetyl groups from lysine residues on histone proteins, resulting in tighter DNA–histone interactions and reduced gene transcription. In many cancers, abnormal HDAC activity contributes to the silencing of tumor suppressor genes. Inhibiting these enzymes can restore expression of genes involved in cell cycle regulation and apoptosis, making HDACs important therapeutic targets. Tucidinostat was developed as a selective inhibitor of class I and class II HDAC enzymes. Its chemical structure contains an aromatic cap group, a linker region, and a hydroxamic acid functional group, which is responsible for binding to the zinc ion in the active site of HDAC enzymes. This coordination inhibits enzymatic activity by blocking access to the substrate acetyl-lysine residues on histones and other proteins. The discovery of tucidinostat is part of broader efforts in medicinal chemistry to develop epigenetic drugs with improved selectivity and safety profiles compared with earlier HDAC inhibitors. Earlier compounds demonstrated that HDAC inhibition could have antitumor effects, but challenges such as toxicity and limited selectivity drove the development of more refined molecules. Tucidinostat was designed to balance potency with reduced off-target effects. In biological terms, tucidinostat increases histone acetylation levels within cells, leading to a more open chromatin structure and increased transcription of genes involved in cell cycle arrest and apoptosis. It can also affect non-histone proteins that are substrates for HDACs, influencing additional cellular processes such as protein stability and signal transduction. These combined effects contribute to its anticancer activity. Clinically, tucidinostat is used for the treatment of relapsed or refractory peripheral T-cell lymphoma and has also been studied in other hematologic and solid tumors. It is typically administered orally and is often used in patients who have not responded to conventional chemotherapy. Its approval in certain regions was based on clinical trials demonstrating antitumor activity and manageable safety profiles in specific patient populations. Pharmacokinetically, tucidinostat is absorbed after oral administration and undergoes hepatic metabolism. Its metabolites and parent compound are eliminated primarily through fecal and renal routes. The drug’s dosing schedule is designed to balance therapeutic efficacy with the management of adverse effects. Adverse effects associated with tucidinostat are related to its impact on rapidly dividing normal cells and epigenetic regulation. These may include hematologic toxicities such as thrombocytopenia and neutropenia, as well as gastrointestinal symptoms and fatigue. Careful monitoring of blood counts is typically required during treatment. Overall, tucidinostat is a targeted epigenetic therapy that inhibits histone deacetylases to restore normal gene expression patterns in cancer cells. Its discovery reflects advances in understanding the role of epigenetic regulation in oncology, and its clinical use represents part of a growing class of drugs that modulate chromatin structure to treat malignancies. References 2026. Novel agents and therapeutic advances in T cell lymphoma. International Journal of Hematology. DOI: 10.1007/s12185-026-04165-7 2026. Differential transcriptomic modulation by histone deacetylase inhibitor SAHA in LUAD and LUSC. Clinical Epigenetics. DOI: 10.1186/s13148-026-02051-x 2011. Chidamide (CS055/HBI-8000): a new histone deacetylase inhibitor of the benzamide class with antitumor activity and the ability to enhance immune cell-mediated tumor cell cytotoxicity. Cancer Chemotherapy and Pharmacology. DOI: 10.1007/s00280-011-1766-x |
| Market Analysis Reports |