Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Beijing Mesochem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.mesochem.com | |||
 | +86 (10) 5786-2036 57862181 67374028 +86 13366977697 | |||
 | +86 (10) 5786-2181 | |||
 | sales@mesochem.com huafenginfo@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Lianyungang Guike Pharmaceutical Co., ltd. | China | |||
|---|---|---|---|---|
 | www.guikepharm.com | |||
 | +86 (518) 8607-0808 8607-0855 | |||
 | +86 (518) 8607-0852 | |||
 | sale@guikepharm.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Suzhou Bichal Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bichal.com | |||
 | +86 (512) 6805-1130 | |||
 | +86 (512) 6805-2322 | |||
 | info@bichal.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Synthland Limited | China | |||
|---|---|---|---|---|
 | www.synthlandpharm.com | |||
 | +86 (755) 8333-1328 | |||
 | info@synthlandpharm.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Porton Corporation | China | |||
|---|---|---|---|---|
 | www.porton.cn | |||
 | +86 (23) 6763-5888 | |||
 | +86 (23) 6703-8513 / 6763-5889 | |||
 | porton@porton.cn | |||
| Chemical manufacturer since 2005 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Kemprotec Limited | UK | |||
|---|---|---|---|---|
 | www.kemprotec.com | |||
 | +44 (1642) 591-764 | |||
 | +44 (1845) 550-001 | |||
 | sales@kemprotec.co.uk | |||
| Chemical distributor since 1998 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antineoplastic agents |
|---|---|
| Name | Dexrazoxane |
| Synonyms | (S)-4,4'-(1-Methyl-1,2-ethanediyl)bis-2,6-piperazinedione |
| Molecular Structure | 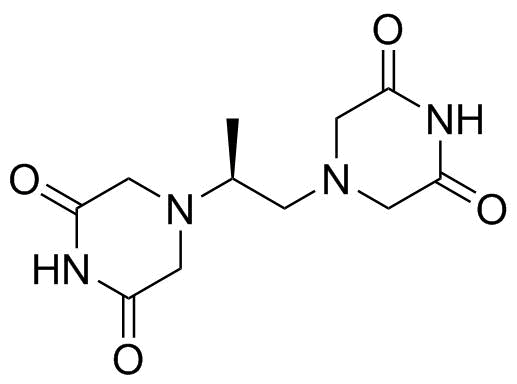 |
| Molecular Formula | C11H16N4O4 |
| Molecular Weight | 268.27 |
| CAS Registry Number | 24584-09-6 |
| EC Number | 635-584-1 |
| SMILES | C[C@@H](CN1CC(=O)NC(=O)C1)N2CC(=O)NC(=O)C2 |
| Density | 1.3$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 531.5$+/-$50.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 275.3$+/-$30.1 $degree$C (Calc.)* |
| Solubility | DMSO 60 mg/mL, Water 60 mg/mL (Expl.) |
| Index of refraction | 1.54 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Dexrazoxane is a synthetic small-molecule drug used primarily as a cardioprotective agent in patients receiving anthracycline chemotherapy and as a treatment for anthracycline extravasation injury. It belongs to the class of bisdioxopiperazine compounds and is structurally related to EDTA-like chelating agents. The development of dexrazoxane was driven by the clinical need to reduce the dose-limiting cardiotoxicity associated with anthracycline drugs such as doxorubicin. Anthracyclines are highly effective anticancer agents, but their use is limited by cumulative, irreversible damage to cardiac tissue. This toxicity is largely associated with iron-dependent oxidative stress mechanisms. Dexrazoxane was designed to mitigate this effect by interfering with iron-mediated free radical formation. Dexrazoxane itself is a prodrug. After administration, it is hydrolyzed intracellularly to an active metabolite that can chelate iron. By binding free iron, it reduces the formation of reactive oxygen species generated through Fenton-type reactions in the presence of anthracyclines. This decreases oxidative damage to cardiac myocytes, which are particularly sensitive to oxidative stress due to relatively low levels of endogenous antioxidant defenses. In addition to its iron-chelating activity, dexrazoxane has been shown to inhibit topoisomerase II. Anthracyclines exert part of their anticancer activity through inhibition of topoisomerase II, but in cardiac cells this interaction can contribute to DNA damage and cytotoxicity. Dexrazoxane modulates this effect by stabilizing topoisomerase II in a manner that reduces DNA double-strand breaks in non-tumor tissues, particularly in the heart. This dual mechanism contributes to its cardioprotective effects. The compound was developed through research into metal chelation therapy and enzyme inhibition. Its structure is based on a cyclic bisdioxopiperazine framework that can undergo hydrolysis to generate chelating species. The parent compound is relatively stable, allowing it to be administered systemically before conversion to active metabolites within cells. Dexrazoxane is administered intravenously and is typically given prior to anthracycline chemotherapy. Its timing is important because it needs to be present in tissues at the time of anthracycline exposure to provide effective cardioprotection. It is also used in the treatment of anthracycline extravasation, where it helps reduce tissue damage caused by accidental leakage of chemotherapy drugs into surrounding tissues. Clinically, dexrazoxane has been shown to reduce the incidence and severity of anthracycline-induced cardiomyopathy, particularly in patients receiving high cumulative doses. Its use allows for more sustained administration of anthracycline chemotherapy in certain cancer treatment protocols, potentially improving oncologic outcomes while reducing cardiac risk. Pharmacokinetically, dexrazoxane is rapidly distributed after intravenous administration and undergoes hydrolysis to active metabolites. It is primarily eliminated through renal excretion. Dose adjustments are required in patients with impaired kidney function to avoid accumulation. Adverse effects associated with dexrazoxane include myelosuppression, gastrointestinal symptoms, and transient elevations in liver enzymes. Because it can affect rapidly dividing cells and topoisomerase activity, there has also been discussion in clinical research about potential interactions with the anticancer efficacy of anthracyclines, although it is generally considered safe and beneficial in appropriate settings. Overall, dexrazoxane is a cardioprotective agent that acts through iron chelation and modulation of topoisomerase II activity to reduce anthracycline-induced cardiac toxicity. Its development represents an important advance in supportive oncology care, enabling safer use of highly effective but cardiotoxic chemotherapeutic agents. References 2026. Remote ischemic conditioning protects against anthracycline cardiotoxicity without impairing its antitumor activity. Basic Research in Cardiology. DOI: 10.1007/s00395-026-01160-1 2026. Dexrazoxane Cardioprotection in pediatric ALL: a historical control cohort study. Cardio-oncology (London, England). DOI: 10.1186/s40959-025-00420-8 2026. Role of Perindopril in Mitigating Doxorubicin’s Vascular Toxicity in a Rat Model. Cardiovascular Toxicology. DOI: 10.1007/s12012-026-10092-0 |
| Market Analysis Reports |