Online Database of Chemicals from Around the World
| CAS: 30817-90-4 Product: 4,4'-(Cyclohexane-1,1-diyl)bis(2-aminophenol) No suppliers available. |
| Classification | Natural product >> Natural phenols |
|---|---|
| Name | 4,4'-(Cyclohexane-1,1-diyl)bis(2-aminophenol) |
| Synonyms | 2-amino-4-[1-(3-amino-4-hydroxyphenyl)cyclohexyl]phenol |
| Molecular Structure | 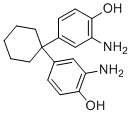 |
| Molecular Formula | C18H22N2O2 |
| Molecular Weight | 298.38 |
| CAS Registry Number | 30817-90-4 |
| SMILES | C1CCC(CC1)(C2=CC(=C(C=C2)O)N)C3=CC(=C(C=C3)O)N |
| Density | 1.3$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 518.8$+/-$50.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 267.6$+/-$30.1 $degree$C (Calc.)* |
| Index of refraction | 1.68 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
4,4'-(Cyclohexane-1,1-diyl)bis(2-aminophenol) is a multifunctional aromatic compound in which two 2-aminophenol units are linked through a cyclohexane bridge at the 4-positions of the phenyl rings. Compounds of this type are important intermediates in polymer chemistry, coordination chemistry, and materials science because they combine rigid aromatic systems with flexible alicyclic linkages and multiple reactive functional groups. The presence of both amino and hydroxyl substituents on each aromatic ring enables extensive hydrogen bonding and diverse chemical transformations. The molecular structure consists of a central cyclohexane ring substituted at the 1,1-positions by two phenyl groups. Each phenyl ring carries an amino group at the 2-position and a hydroxyl group also at the ortho position relative to the linkage. This arrangement provides four functional groups per molecule, two amino and two hydroxyl, which can participate in condensation reactions, coordination to metal centers, or formation of polymeric networks. The cyclohexane core introduces conformational flexibility compared to fully aromatic systems, which can influence solubility and processing behavior in material applications. The synthesis of 4,4'-(cyclohexane-1,1-diyl)bis(2-aminophenol) typically involves condensation of cyclohexanone or related derivatives with substituted nitrophenols, followed by reduction of nitro groups to amino groups. Careful control of reaction conditions ensures substitution at the para positions of the aromatic rings and preservation of the hydroxyl functionality. The final product is generally obtained as a stable solid that can be purified and used as an intermediate in further chemical reactions. In polymer chemistry, compounds containing both amino and hydroxyl groups are valuable monomers for the preparation of high-performance materials. The amino groups can react with diacid chlorides or dianhydrides to form polyamides or polyimides, while the hydroxyl groups can participate in additional crosslinking or modification reactions. The cyclohexane linkage can improve flexibility and reduce brittleness in the resulting polymers, while the aromatic rings provide thermal stability and mechanical strength. Such materials are used in coatings, adhesives, and advanced composites. In coordination chemistry, 2-aminophenol derivatives are known to act as ligands capable of binding metal ions through both nitrogen and oxygen atoms. The presence of two such units in a single molecule allows the formation of chelating or bridging complexes with transition metals. These complexes can exhibit catalytic activity or useful electronic and optical properties, making the compound relevant in catalysis and materials research. Beyond these applications, 4,4'-(cyclohexane-1,1-diyl)bis(2-aminophenol) serves as a versatile intermediate in organic synthesis. The amino groups can undergo acylation, alkylation, or diazotization, while the hydroxyl groups can be converted into ethers or esters. This versatility enables the preparation of a wide range of derivatives with tailored properties for specific applications in chemical research and material development. Overall, 4,4'-(cyclohexane-1,1-diyl)bis(2-aminophenol) is a multifunctional compound combining aromatic reactivity, hydrogen-bonding capability, and a flexible cyclohexane core. Its structural features make it valuable for polymer synthesis, coordination chemistry, and the development of advanced materials and functional molecules. References 2024. Polymer, negative photosensitive resin composition, patterning process, method for forming cured film, interlayer insulating film, surface protective film, and electronic component. EP-4636011-A1. URL: https://patents.google.com/patent/EP4636011A1/en 2024. Negative photosensitive resin composition, patterning process, method for forming cured film, interlayer insulating film, surface protective film, and electronic component. EP-4636485-A1. URL: https://patents.google.com/patent/EP4636485A1/en 2023. Polymer, Positive And Negative Photosensitive Resin Compositions, Patterning Process, Method For Forming Cured Film, Interlayer Insulating Film, Surface Protective Film, And Electronic Component. US-2025147420-A1. URL: https://patents.google.com/patent/US2025147420A1/en |
| Market Analysis Reports |