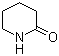
2-Piperidone, also known as δ-valerolactam, is a six-membered lactam with the molecular formula C5H9NO. It is a versatile organic compound containing an amide functional group within a cyclic structure, making it a critical intermediate in the synthesis of various pharmaceuticals, agrochemicals, and polymers. Its properties include a solid-state at room temperature, high solubility in water and organic solvents, and reactivity in ring-opening and substitution reactions.
The discovery of 2-piperidone can be traced to early studies in the field of lactams during the late 19th and early 20th centuries. Researchers investigating nitrogen-containing heterocycles identified 2-piperidone as a potential building block for more complex molecules. Advances in synthetic chemistry allowed for its efficient preparation through routes such as cyclization of 5-aminovaleric acid or oxidative cleavage of caprolactam.
In the pharmaceutical industry, 2-piperidone plays a significant role as a precursor for the synthesis of drugs targeting neurological and metabolic disorders. Its cyclic structure serves as a core for many bioactive compounds, including anticonvulsants, analgesics, and muscle relaxants. The lactam ring provides a scaffold for further functionalization, enabling the creation of structurally diverse molecules with tailored pharmacological properties.
The compound also finds applications in the development of agrochemicals, where it is utilized as a key intermediate in the production of herbicides, fungicides, and insecticides. Its reactivity allows for the introduction of substituents that enhance biological activity, stability, and selectivity. This versatility has made 2-piperidone a valuable component in the design of chemicals aimed at improving crop yields and controlling pests.
One of the most notable industrial applications of 2-piperidone is in polymer chemistry. It serves as a monomer for the production of polyamides, particularly nylon-6, which is widely used in textiles, engineering plastics, and automotive components. The ring-opening polymerization of 2-piperidone yields high-performance materials with excellent mechanical strength, thermal resistance, and durability.
Research continues to explore the potential of 2-piperidone in green chemistry and sustainable materials. Efforts focus on developing bio-based synthesis methods and recycling processes to reduce environmental impact. Additionally, its use in advanced drug delivery systems and biodegradable polymers highlights its growing importance in emerging technologies.
References
2024. Identification of avaC from Human Gut Microbial Isolates that Converts 5AVA to 2-Piperidone. Journal of microbiology (Seoul, Korea), 62(5).
DOI: 10.1007/s12275-024-00141-0
2024. One-pot formation of six bonds: pseudo-six-component diastereoselective synthesis of pyridinium-containing piperidin-2-ones. Russian Chemical Bulletin, 73(5).
DOI: 10.1007/s11172-024-4242-6
2024. Synthesis, anticancer evaluation, preliminary mechanism study of novel 1, 2, 3-triazole-piperlongumine derivatives. Molecular Diversity, 28(6).
DOI: 10.1007/s11030-024-11021-5
|

























































 GHS02;GHS05;GHS07 Danger Details
GHS02;GHS05;GHS07 Danger Details