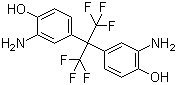
2,2-Bis(3-amino-4-hydroxyphenyl)hexafluoropropane is an aromatic diamine compound in which two 3-amino-4-hydroxyphenyl groups are linked to a central hexafluoropropane moiety. This molecular arrangement combines nucleophilic amino groups and hydroxyl groups with a highly electron-withdrawing and sterically bulky trifluoromethyl-substituted carbon center. The compound is primarily used as a monomer in the synthesis of high-performance polymers, especially epoxy resins and polyimides, where thermal stability, chemical resistance, and mechanical strength are critical.
The molecular structure features a central carbon atom substituted with two trifluoromethyl groups and connected to two para-hydroxylated, meta-amino-substituted benzene rings. The amino groups provide reactive sites for condensation and polymerization reactions, while the hydroxyl groups allow etherification, crosslinking, or further chemical modifications. The hexafluoropropane core introduces rigidity, electron-withdrawing character, and steric hindrance, which enhance the thermal stability and chemical resistance of resulting polymers.
Synthesis of 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane typically starts from bisphenol A derivatives or hexafluoropropane precursors, which are nitrated and then reduced to introduce the amino groups while preserving the hydroxyl functionality. Reaction conditions are carefully controlled to avoid oxidation or side reactions that could compromise the purity and reactivity of the diamine. High-purity products are essential for the preparation of advanced polymer materials.
One of the main applications of this compound is in the production of high-performance epoxy resins. Polymers derived from this diamine exhibit exceptional thermal and chemical resistance, mechanical strength, and low moisture absorption, making them suitable for aerospace, electronics, and automotive applications. These epoxy resins are used in circuit boards, structural adhesives, protective coatings, and composite materials that require durability under extreme temperature and chemical exposure.
In addition to epoxy resins, 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane is employed in polyimide synthesis, where its bifunctional nature allows the formation of thermally stable, high-strength films and fibers. Its combination of nucleophilic amino groups, hydroxyl functionality, and the fluorinated central core also enables its use as a building block in specialty chemicals, dyes, and functionalized aromatic compounds.
Overall, 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane is a versatile, thermally stable, and chemically robust aromatic diamine. Its amino and hydroxyl groups, combined with the electron-withdrawing hexafluoropropane core, make it a key intermediate for high-performance polymers, epoxy resins, polyimides, and specialty chemical synthesis.
References
2024. Synthesis of UV-resistant and colorless polyimide films for optoelectrical applications. npj Materials Degradation.
DOI: 10.1038/s41529-023-00422-w
2023. Polysilsesquioxane-PBO Wave-Transparent Composite Paper with Excellent Mechanical Properties and Ultraviolet Aging Resistance. Advanced Fiber Materials.
DOI: 10.1007/s42765-023-00327-y
2023. A Novel Naphthylidene-diimine Chemosensor for Selective Colorimetric and Fluorometric Detection of Al3+ and CN− Ions. Journal of Fluorescence.
DOI: 10.1007/s10895-023-03368-z
|


















































 GHS07 Warning Details
GHS07 Warning Details