Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Hohance Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hohance.com | |||
 | +86 (21) 3111-5312 | |||
 | +86 (21) 3111-5317 | |||
 | info@hohance.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Qinba Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shangfluoro.com | |||
 | +86 (21) 6517-0832 +86 17301729617 | |||
 | +86 (21) 6517-0832 | |||
 | shangfluoro@hotmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Classification | Organic raw materials >> Organic fluorine compound |
|---|---|
| Name | 3,3-Dimethyl-1-(trifluoromethyl)-1,2-benziodoxole |
| Synonyms | Togni's Reagent |
| Molecular Structure | 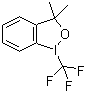 |
| Molecular Formula | C10H10F3IO |
| Molecular Weight | 330.08 |
| CAS Registry Number | 887144-97-0 |
| EC Number | 626-754-6 |
| SMILES | CC1(C2=CC=CC=C2I(O1)C(F)(F)F)C |
| Melting point | 75 - 79 $degree$C (Expl.) |
|---|---|
| Hazard Symbols |   GHS02;GHS07 DangerGHS02; Details GHS02;GHS07 DangerGHS02; Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H228-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P210-P240-P241-P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3,3-Dimethyl-1-(trifluoromethyl)-1,2-benziodoxole is a hypervalent iodine compound in which a benzene ring is fused to a five-membered iodine-containing heterocycle, with a trifluoromethyl group attached to the iodine center and two methyl groups at the 3-position of the heterocycle. This molecular structure combines the electrophilic reactivity of hypervalent iodine with the electron-withdrawing properties of the trifluoromethyl group, making it a valuable reagent in organic synthesis. The molecular structure consists of a benziodoxole core, where the iodine atom is in a +3 oxidation state, forming a three-center, four-electron bond characteristic of λ3-iodanes. The trifluoromethyl substituent increases the electrophilicity of the iodine, enhancing its ability to transfer CF3 groups or act as an oxidizing agent. The two methyl groups at the 3-position stabilize the heterocyclic ring and influence the steric environment around the reactive iodine center. Synthesis of 3,3-dimethyl-1-(trifluoromethyl)-1,2-benziodoxole typically involves oxidation of iodoarenes to form the hypervalent iodine intermediate, followed by trifluoromethylation using reagents such as Ruppert–Prakash reagents or other CF3-transfer reagents. The resulting compound is usually isolated as a stable solid that can be handled under ambient conditions, though care must be taken due to its oxidizing properties. This compound finds application primarily as a trifluoromethylating reagent in organic synthesis. It enables the introduction of CF3 groups onto nucleophilic centers, such as enolates, aromatics, and heteroaromatics, under mild conditions. The presence of the hypervalent iodine allows selective and efficient transfer of the trifluoromethyl group, which is important in the synthesis of pharmaceuticals, agrochemicals, and materials with enhanced chemical and physical properties. In addition to trifluoromethylation, the hypervalent iodine center in this molecule can serve as an oxidizing agent for various transformations, including oxidative rearrangements, cyclizations, and functional group interconversions. Its stability and reactivity make it a convenient alternative to more hazardous or less selective oxidants. Overall, 3,3-dimethyl-1-(trifluoromethyl)-1,2-benziodoxole is a versatile hypervalent iodine reagent that combines the electrophilic nature of λ3-iodanes with the electron-withdrawing effect of a trifluoromethyl group. Its structural and electronic features make it a valuable tool for trifluoromethylation and oxidative transformations in modern organic synthesis. References 2023. Zirconium Halide Complexes: Synthesis, Structure, and Practical Applications. Reviews and Advances in Chemistry. DOI: 10.1134/s2634827623700162 2022. Recent Advances in Transition-Metal-Catalyzed Asymmetric Functionalization of Enamides. Synthesis. DOI: 10.1055/a-1892-5473 2022. Catalytic Asymmetric Construction of CF3-Substituted Chiral sp3 Carbon Centers. Synthesis. DOI: 10.1055/a-1833-8813 |
| Market Analysis Reports |