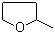
2-Methyltetrahydrofuran (2-MeTHF) is a five-membered cyclic ether with the molecular formula C5H10O. It is structurally similar to tetrahydrofuran (THF), differing by the presence of a methyl group at the 2-position of the ring. This relatively small structural change results in several differences in physical and chemical properties, making 2-MeTHF a valuable solvent in both laboratory and industrial settings.
The compound occurs naturally in trace amounts in some fermentation products and has been identified in bio-oils derived from lignocellulosic biomass. However, 2-methyltetrahydrofuran is primarily produced synthetically. It can be obtained via the catalytic hydrogenation of furfural or levulinic acid derivatives, often derived from renewable feedstocks such as agricultural waste. This route makes 2-MeTHF a more sustainable alternative to petroleum-based solvents.
The adoption of 2-MeTHF as a solvent has increased over the past two decades, particularly in the field of green chemistry. Compared to THF, 2-MeTHF has a higher boiling point (approximately 80 °C), is less hygroscopic, and exhibits improved stability against peroxide formation. It also shows better partitioning in biphasic reactions due to its partial immiscibility with water, which can be advantageous in extraction and reaction processes involving aqueous and organic phases.
In organic synthesis, 2-MeTHF serves as a medium for a wide range of reactions, including Grignard reactions, reductions, and organolithium chemistry. Its compatibility with strong bases and organometallic reagents makes it a preferred solvent in pharmaceutical and fine chemical manufacturing. In particular, 2-MeTHF is used in lithium aluminum hydride and borane reductions, as it is more stable and easier to handle than THF under similar conditions.
The compound has found applications in catalytic and enantioselective reactions, especially in cases where greener solvents are required to meet regulatory or environmental standards. Due to its favorable environmental profile, 2-MeTHF is included on several green solvent selection guides, such as those published by pharmaceutical companies aiming to reduce solvent waste and toxicity.
In battery and energy research, 2-MeTHF has been investigated as a solvent or co-solvent in lithium-ion battery electrolytes. Its electrochemical properties and compatibility with lithium salts make it a candidate for improving battery performance, although its use in this field is still under evaluation.
2-Methyltetrahydrofuran is typically available as a colorless liquid with an ether-like odor. It is flammable and must be handled with appropriate precautions, including storage under inert atmosphere to minimize the formation of peroxides during long-term storage. Like other ethers, it should be tested periodically for peroxide formation and discarded if peroxides are detected above safe thresholds.
From a toxicological perspective, 2-MeTHF is considered to have low acute toxicity and is less volatile than THF, reducing the risk of inhalation exposure. Its classification and handling guidelines are generally similar to other low molecular weight ethers. It is important, however, to follow standard industrial hygiene practices and use appropriate personal protective equipment when handling the compound.
In summary, 2-methyltetrahydrofuran is a versatile, bio-based solvent that offers several advantages over traditional ethers, including improved stability, reduced water miscibility, and environmental benefits. Its growing role in synthetic chemistry and sustainable manufacturing highlights its value in modern chemical processes.
References
2022. Stabilization of intermediate spin states in mixed-valent diiron dichalcogenide complexes. Nature Chemistry, 14(2).
DOI: 10.1038/s41557-021-00853-5
2011. Direct Hydrocyclization of Biomass‐Derived Levulinic Acid to 2‐Methyltetrahydrofuran over Nanocomposite Copper/Silica Catalysts. ChemSusChem, 4(11).
DOI: 10.1002/cssc.201100380
2015. One‐Pot 2‐Methyltetrahydrofuran Production from Levulinic Acid in Green Solvents Using Ni‐Cu/Al2O3 Catalysts. ChemSusChem, 8(17).
DOI: 10.1002/cssc.201500671
|










































































 GHS02;GHS05;GHS07 Danger Details
GHS02;GHS05;GHS07 Danger Details