Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Haimen RuiYi Medical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ruiyitech.cn | |||
 | +86 (21) 5471-1706 | |||
 | +86 (21) 5471-1707 | |||
 | sales@ruiyitech.com zyj3625@msn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Gou Hebei Chemicals | India | |||
|---|---|---|---|---|
 | gouhebeichems.store | |||
 | +86 15874272473 | |||
 | info@gouhebeichems.store | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Nantong Zhonghe Chemical New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ntzhhg.com | |||
 | +86 13003551299 | |||
 | 2369399482@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Telly Chemicals Pvt | China | |||
|---|---|---|---|---|
 | tellychemicals.ecer.com | |||
 | +852 52919765 | |||
 | +852 52919765 | |||
 | tellychemicals@protonmail.com | |||
 | Skype Chat | |||
 | WhatsApp:+61 485956223 | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2024 | ||||
| AccuStandard Inc. | USA | |||
|---|---|---|---|---|
 | www.accustandard.com | |||
 | +1 (203) 786-5290 | |||
 | +1 (203) 786-5287 | |||
 | orders@accustandard.com | |||
| Chemical manufacturer since 1986 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Methyl ester compound |
|---|---|
| Name | 1,4-Benzenedicarboxylic acid 1,4-dimethyl ester |
| Synonyms | Terephthalic acid dimethyl ester; DMT; Dimethyl 1,4-benzenedicarboxylate; Dimethyl p-benzenedicarboxylate; Dimethyl p-phthalate; Dimethyl terephthalate; Methyl 4-(carbomethoxy)benzoate; Methyl p-(methoxycarbonyl)benzoate; NSC 3503 |
| Molecular Structure | 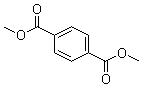 |
| Molecular Formula | C10H10O4 |
| Molecular Weight | 194.18 |
| CAS Registry Number | 120-61-6 |
| EC Number | 204-411-8 |
| SMILES | COC(=O)C1=CC=C(C=C1)C(=O)OC |
| Density | 1.175 |
|---|---|
| Melting point | 139-140 °C |
| Boiling point | 288 °C |
| Flash point | 146 °C |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H303-H316-H320-H372-H335-H401 Details | ||||||||||||||||||||||||||||
| Safety Statements | P501-P273-P260-P270-P271-P264-P312-P337+P313-P305+P351+P338-P332+P313-P304+P340+P312-P403+P233-P405 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
1,4-Dimethyl 1,4-benzenedicarboxylate, commonly known as dimethyl terephthalate (DMT), is a key chemical in the modern polymer industry. DMT was first synthesized in the 1940s during research on aromatic esters. Its structure consists of a benzene ring with two ester groups located in the para (1,4) positions, which provide the rigidity and symmetry required for polymer applications. Direct esterification of terephthalic acid with methanol produces DMT, making it a relatively readily available compound for industrial use. The main application of DMT is in the production of PET. PET is widely used in the manufacture of plastic bottles, packaging materials, and synthetic fibers such as polyester. The polymerization process involves the reaction of DMT with ethylene glycol to form PET through transesterification and polycondensation reactions. The process produces a high-strength, versatile plastic that is essential for a variety of consumer and industrial products. PET's strength, clarity, and moisture resistance make it ideal for beverage bottles, food packaging, and synthetic fibers. DMT is also used in the manufacture of other thermoplastics and engineering resins, where the ester groups contribute to tensile strength, chemical resistance, and dimensional stability. In addition to PET, DMT is used to produce polybutylene terephthalate (PBT), which is used in automotive parts and electrical insulators due to its high thermal stability. DMT is also used to produce liquid crystal polymers (LCP), which are used in electronic displays and components due to their excellent mechanical properties and heat resistance. DMT is used to make engineering resins, films, and coatings, where its durability and transparency are its strengths. The ester groups in DMT promote strong intermolecular bonding, resulting in materials with excellent mechanical and thermal properties. While DMT and its derivatives are essential to modern life, their widespread use raises environmental concerns. PET is not biodegradable, contributing to a serious plastic waste problem. However, recycling PET into new products is a sustainable solution, and ongoing research aims to enhance the recycling process and develop biodegradable alternatives. References 1955. Macromolecular compounds. Bulletin of the Academy of Sciences of the USSR, Division of Chemical Science. DOI: 10.1007/bf01172125 2024. Synthesis and Properties Study of PESF-b-PBIT White Fluorescent Copolyesters. Arabian Journal for Science and Engineering. DOI: 10.1007/s13369-024-09811-y 2024. Effects of Copolymer Structure on Enzyme-Catalyzed Polyester Recycling. Journal of Polymers and the Environment. DOI: 10.1007/s10924-024-03223-7 |
| Market Analysis Reports |