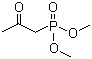
Dimethyl acetylmethylphosphonate is an organophosphorus compound in which a phosphonate group is esterified with two methyl groups and linked to an acetylmethyl moiety. The molecule combines a phosphonate functional group with a carbonyl-containing side chain, providing both nucleophilic and electrophilic reactivity. This structural arrangement makes it a versatile intermediate in organic synthesis, particularly in the preparation of phosphorylated derivatives, reagents for carbon–carbon bond formation, and organophosphorus compounds.
The molecular structure consists of a phosphorus atom double-bonded to an oxygen atom and bonded to two methoxy groups, forming the dimethyl phosphonate ester. The phosphorus is also attached to an acetylmethyl group (–CH2–CO–CH3), which contains a reactive carbonyl functional group. The presence of the carbonyl adjacent to the phosphonate allows for nucleophilic addition, enolate formation, and other condensation reactions, enabling the compound to participate in a wide range of chemical transformations.
Synthesis of dimethyl acetylmethylphosphonate generally involves the reaction of acetylmethyl halides or esters with trialkyl phosphites under controlled conditions. The reaction follows the Michaelis–Arbuzov pathway, where the phosphorus nucleophile displaces a leaving group on the carbon atom to form the phosphonate ester. Reaction parameters such as temperature, solvent, and stoichiometry are optimized to ensure high yield and purity. The resulting dimethyl acetylmethylphosphonate is a stable liquid suitable for further chemical reactions.
This compound is widely used in organic synthesis as a precursor to α-phosphono ketones, β-ketophosphonates, and other phosphorylated compounds. Its phosphonate moiety allows for the Horner–Wadsworth–Emmons (HWE) reaction, enabling the formation of carbon–carbon double bonds with aldehydes and ketones to generate substituted alkenes with high stereoselectivity. Such reactions are valuable in the synthesis of complex molecules, including natural products, pharmaceuticals, and functional materials.
In addition, dimethyl acetylmethylphosphonate serves as an intermediate in the preparation of ligands, catalysts, and biologically active organophosphorus compounds. The combination of the phosphonate ester and acetyl functional group provides multiple reactive sites that can be chemically modified to tune the electronic and steric properties of derived compounds. Its chemical versatility has made it a common reagent in research laboratories and industrial applications.
Overall, dimethyl acetylmethylphosphonate is a multifunctional organophosphorus compound with important applications in organic synthesis, chemical intermediates, and the preparation of phosphorylated derivatives. Its combination of a phosphonate ester, carbonyl functionality, and reactive methylene group provides flexibility for diverse chemical transformations, making it a valuable building block for the synthesis of complex molecules and functional materials.
References
2024. Total Synthesis of Amycolamicin. Modern Natural Product Synthesis.
DOI: 10.1007/978-981-97-1619-7_2
2022. Galiellalactone: a review on synthetic strategies and tactics. Chemical Papers.
DOI: 10.1007/s11696-022-02630-1
2021. Applications of the Horner–Wadsworth–Emmons Olefination in Modern Natural Product Synthesis. Synthesis.
DOI: 10.1055/a-1493-6331
|



































 GHS07;GHS08 Warning Details
GHS07;GHS08 Warning Details